Genetically Tailored Porcine Disease Models
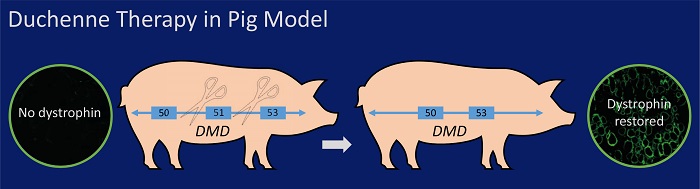
Duchenne muscular dystrophy (DMD) is the most common hereditary muscular disease among children, often forcing victims into the wheelchair before the age of twelve and reducing life expectancies. A tailored pig model for DMD (Klymiuk et al., Hum Mol Genet 2013) was instrumental for the development of a gene therapy that may provide relief for those suffering from DMD (Moretti et al., Nat Med 2020). The same model has recently been for validating a novel non-invasive procedure for monitoring disease progression (Regensburger et al., Nat Med 2019).
Research into disease mechanisms is an important basis for the development of novel, targeted therapies. The path from disease-oriented basic research towards clinical application in patients (= translational medicine) is however long and cost-intensive. Suitable animal models, which allow predictions on the efficacy and safety of novel therapies are inevitable in this process. Rodent models are most widely used, but are often limited in the resemblance of human disease mechanisms and phenotypes. Therefore, effects of new treatments in humans are sometimes difficult to predict on the basis of findings in rodents. Thus, large animal models that mimic anatomical and physiological parameters of humans more closely are additionally needed. For a number of reasons, the pig is a relevant translational animal model. Due to the establishment of efficient and precise techniques for genetic modification of pigs, it is possible to generate tailored pig models, which mimic human disease mechanisms on a molecular and functional level.
As the Large Animal Platform of the German Center for Diabetes Research (DZD; https://www.dzd-ev.de/), we generated genetically diabetic or prediabetic pig models, perform treatment studies, and analyse diabetic complications and organ cross-talk using multi-omics designs. In addition, we generated several reporter pig lines for studying pancreatic islet cell biology.
Another focus is the generation of pig models for monogenic diseases, including cystic fibrosis (CF), Duchenne muscular dystrophy (DMD) and Laron syndrome (LS).
Highlights
- CRISPR/Cas based therapy evaluated in DMD pig model (Moretti et al., Nat Med 2020)
- Multispectral optoacoustic tomography (MSOT) validated as imaging biomarker for DMD (Regensburger et al., Nat Med 2019)
- First multi-omics study of functional changes of the liver in insulin-deficient diabetes mellitus (Backmann et al., Mol Metab 2019)
- Novel pig model for gestational diabetes established (Renner et al., Mol Metab 2019)
- Growth hormone receptor deficient pigs show the hallmarks of Laron syndrome (Hinrichs et al., Mol Metab 2018)
- The Munich MIDY Pig Biobank as a unique resource for studying organ cross-talk in diabetes mellitus (Blutke et al., Mol Metab 2017)
- INSC49Y pigs – a model for mutant INS gene induced diabetes of youth (MIDY) (Renner et al., Diabetes 2013)
- First pig model for Duchenne muscular dystrophy (Klymiuk et al., Hum Mol Genet 2013)
- GIPRdn transgenic pigs resembling a prediabetic condition (Renner et al., Diabetes 2010) were used for biomarker discovery (Renner et al., Diabetes 2012) and treatment studies (Streckel et al., J Tanslat Med 2015)
Publications
2023
Gong S, Tetti M, Kemter E, Peitzsch M, Mulatero P, Bidlingmaier M, Eisenhofer G, Wolf E, Reincke M, Williams TA. TSPAN12 (Tetraspanin 12) Is a Novel Negative Regulator of Aldosterone Production in Adrenal Physiology and Aldosterone-Producing Adenomas. Hypertension. 2023 Feb;80(2):440-450. PMID: 36458545
Pauly V, Vlcek J, Zhang Z, Hesse N, Xia R, Bauer J, Loy S, Schneider S, Renner S, Wolf E, Kääb S, Schüttler D, Tomsits P, Clauss S. Effects of Sex on the Susceptibility for Atrial Fibrillation in Pigs with Ischemic Heart Failure. Cells. 2023 Mar 23;12(7):973. doi: 10.3390/cells12070973. PMID: 37048048; PMCID: PMC10093477
Schilloks MC, Giese IM, Hinrichs A, Korbonits L, Hauck SM, Wolf E, Deeg CA. Effects of GHR Deficiency and Juvenile Hypoglycemia on Immune Cells of a Porcine Model for Laron Syndrome. Biomolecules. 2023 Mar 26;13(4):597. doi: 10.3390/biom13040597. PMID: 37189345; PMCID: PMC10135491
Shashikadze B, Valla L, Lombardo SD, Prehn C, Haid M, Riols F, Stöckl JB, Elkhateib R, Renner S, Rathkolb B, Menche J, Hrabĕ de Angelis M, Wolf E, Kemter E, Fröhlich T. Maternal hyperglycemia induces alterations in hepatic amino acid, glucose and lipid metabolism of neonatal offspring: Multi-omics insights from a diabetic pig model. Mol Metab. 2023 Jul 4;75:101768. doi: 10.1016/j.molmet.2023.101768. Epub ahead of print. PMID: 37414142
Shashikadze B, Franzmeier S, Hofmann I, Kraetzl M, Flenkenthaler F, Blutke A, Fröhlich T, Wolf E, Hinrichs A. Structural and proteomic repercussions of growth hormone receptor deficiency on the pituitary gland: Lessons from a translational pig model. J Neuroendocrinol. 2023 Apr 15:e13277. doi: 10.1111/jne.13277. Epub ahead of print. PMID: 37160285.
Stirm M, Shashikadze B, Blutke A, Kemter E, Lange A, Stöckl JB, Jaudas F, Laane L, Kurome M, Keßler B, Zakhartchenko V, Bähr A, Klymiuk N, Nagashima H, Walter MC, Wurst W, Kupatt C, Fröhlich T, Wolf E. Systemic deletion of DMD exon 51 rescues clinically severe Duchenne muscular dystrophy in a pig model lacking DMD exon 52. Proc Natl Acad Sci U S A. 2023 Jul 18;120(29):e2301250120. doi: 10.1073/pnas.2301250120. Epub 2023 Jul 10. PMID: 37428903
2022
Donandt T, Hintze S, Krause S, Wolf E, Schoser B, Walter MC, Meinke P. Isolation and Characterization of Primary DMD Pig Muscle Cells as an In Vitro Model for Preclinical Research on Duchenne Muscular Dystrophy. Life (Basel). 2022 Oct 21;12(10):1668. PMID: 36295103
Flenkenthaler F, Ländström E, Shashikadze B, Backman M, Blutke A, Philippou-Massier J, Renner S, Hrabe de Angelis M, Wanke R, Blum H, Arnold GJ, Wolf E, Fröhlich T. Differential Effects of Insulin-Deficient Diabetes Mellitus on Visceral vs. Subcutaneous Adipose Tissue-Multi-omics Insights From the Munich MIDY Pig Model. Front Med (Lausanne). 2021 Nov 23;8:751277. PMID: 34888323
Geng R, Knoll J, Harland N, Amend B, Enderle MD, Linzenbold W, Abruzzese T, Kalbe C, Kemter E, Wolf E, Schenk M, Stenzl A, Aicher WK. Replacing Needle Injection by a Novel Waterjet Technology Grants Improved Muscle Cell Delivery in Target Tissues. Cell Transplant. 2022 Jan-Dec;31:9636897221080943. PMID: 35466714
Grotz S, Schäfer J, Wunderlich KA, Ellederova Z, Auch H, Bähr A, Runa-Vochozkova P, Fadl J, Arnold V, Ardan T, Veith M, Santamaria G, Dhom G, Hitzl W, Kessler B, Eckardt C, Klein J, Brymova A, Linnert J, Kurome M, Zakharchenko V, Fischer A, Blutke A, Döring A, Suchankova S, Popelar J, Rodríguez-Bocanegra E, Dlugaiczyk J, Straka H, May-Simera H, Wang W, Laugwitz KL, Vandenberghe LH, Wolf E, Nagel-Wolfrum K, Peters T, Motlik J, Fischer MD, Wolfrum U, Klymiuk N. Early disruption of photoreceptor cell architecture and loss of vision in a humanized pig model of usher syndromes. EMBO Mol Med. 2022 Apr 7;14(4):e14817. PMID: 35254721
Lugarà R, Renner S, Wolf E, Liesegang A, Bruckmaier R, Giller K. Crossbred Sows Fed a Western Diet during Pre-Gestation, Gestation, Lactation, and Post-Lactation Periods Develop Signs of Lean Metabolic Syndrome That Are Partially Attenuated by Spirulina Supplementation. Nutrients. 2022 Aug 30;14(17):3574. PMID: 36079836
Poch CM, Foo KS, De Angelis MT, Jennbacken K, Santamaria G, Bähr A, Wang QD, Reiter F, Hornaschewitz N, Zawada D, Bozoglu T, My I, Meier A, Dorn T, Hege S, Lehtinen ML, Tsoi YL, Hovdal D, Hyllner J, Schwarz S, Sudhop S, Jurisch V, Sini M, Fellows MD, Cummings M, Clarke J, Baptista R, Eroglu E, Wolf E, Klymiuk N, Lu K, Tomasi R, Dendorfer A, Gaspari M, Parrotta E, Cuda G, Krane M, Sinnecker D, Hoppmann P, Kupatt C, Fritsche-Danielson R, Moretti A, Chien KR, Laugwitz KL. Migratory and anti-fibrotic programmes define the regenerative potential of human cardiac progenitors. Nat Cell Biol. 2022 May;24(5):659-671. PMID: 35550611
Shashikadze B, Flenkenthaler F, Stöckl JB, Valla L, Renner S, Kemter E, Wolf E, Fröhlich T. Developmental Effects of (Pre-)Gestational Diabetes on Offspring: Systematic Screening Using Omics Approaches. Genes (Basel). 2021 Dec 15;12(12):1991. PMID: 34946940
Stirm M, Fonteyne LM, Shashikadze B, Stöckl JB, Kurome M, Keßler B, Zakhartchenko V, Kemter E, Blum H, Arnold GJ, Matiasek K, Wanke R, Wurst W, Nagashima H, Knieling F, Walter MC, Kupatt C, Fröhlich T, Klymiuk N, Blutke A, Wolf E. Pig models for Duchenne muscular dystrophy - from disease mechanisms to validation of new diagnostic and therapeutic concepts. Neuromuscul Disord. 2022 Jul;32(7):543-556. PMID: 35659494
Tritschler S, Thomas M, Böttcher A, Ludwig B, Schmid J, Schubert U, Kemter E, Wolf E, Lickert H, Theis FJ. A transcriptional cross species map of pancreatic islet cells. Mol Metab. 2022 Dec;66:101595. PMID: 36113773
2021
Gerst F, Kemter E, Lorza-Gil E, Kaiser G, Fritz AK, Nano R, Piemonti L, Gauder M, Dahl A, Nadalin S, Königsrainer A, Fend F, Birkenfeld AL, Wagner R, Heni M, Stefan N, Wolf E, Häring HU, Ullrich S. The hepatokine fetuin-A disrupts functional maturation of pancreatic beta cells. Diabetologia. 2021 Mar 25. doi: 10.1007/s00125-021-05435-1. Online ahead of print. PMID: 33765181
Giese IM, Schilloks MC, Degroote RL, Weigand M, Renner S, Wolf E, Hauck SM, Deeg CA. Chronic Hyperglycemia Drives Functional Impairment of Lymphocytes in Diabetic INS C94Y Transgenic Pigs. Front Immunol. 2021 Jan 22;11:607473. doi: 10.3389/fimmu.2020.607473. eCollection 2020. PMID: 33552065
Hinrichs A, Renner S, Bidlingmaier M, Kopchick JJ, Wolf E. MECHANISMS IN ENDOCRINOLOGY: Transient juvenile hypoglycemia in growth hormone receptor deficiency - mechanistic insights from Laron syndrome and tailored animal models. Eur J Endocrinol. 2021 Jul 1;185(2):R35-R47. PMID: 34048365
Kalla D, Flisikowski K, Yang K, Sangüesa LB, Kurome M, Kessler B, Zakhartchenko V, Wolf E, Lickert H, Saur D, Schnieke A, Flisikowska T. The Missing Link: Cre Pigs for Cancer Research. Front Oncol. 2021 Oct 8;11:755746. PMID: 34692545
Kemter E, Müller A, Neukam M, Ivanova A, Klymiuk N, Renner S, Yang K, Broichhagen J, Kurome M, Zakhartchenko V, Kessler B, Knoch KP, Bickle M, Ludwig B, Johnsson K, Lickert H, Kurth T, Wolf E, Solimena M. Sequential in vivo labeling of insulin secretory granule pools in INS-SNAP transgenic pigs. Proc Natl Acad Sci U S A. 2021 Sep 14;118(37):e2107665118. PMID: 34508004
Kupatt C, Windisch A, Moretti A, Wolf E, Wurst W, Walter MC. Genome editing for Duchenne muscular dystrophy: a glimpse of the future? Gene Ther. 2021 Feb 2. doi: 10.1038/s41434-021-00222-4. Online ahead of print. PMID: 33531685
Stirm M, Fonteyne LM, Shashikadze B, Lindner M, Chirivi M, Lange A, Kaufhold C, Mayer C, Medugorac I, Kessler B, Kurome M, Zakhartchenko V, Hinrichs A, Kemter E, Krause S, Wanke R, Arnold GJ, Wess G, Nagashima H, Hrabĕ de Angelis M, Flenkenthaler F, Kobelke LA, Bearzi C, Rizzi R, Bähr A, Reese S, Matiasek K, Walter MC, Kupatt C, Ziegler S, Bartenstein P, Fröhlich T, Klymiuk N, Blutke A, Wolf E. A scalable, clinically severe pig model for Duchenne muscular dystrophy. Dis Model Mech. 2021 Dec 1;14(12):dmm049285. PMID: 34796900
Theobalt N, Hofmann I, Fiedler S, Renner S, Dhom G, Feuchtinger A, Walch A, Hrabĕ de Angelis M, Wolf E, Wanke R, Blutke A. Unbiased analysis of obesity related, fat depot specific changes of adipocyte volumes and numbers using light sheet fluorescence microscopy. PLoS One. 2021 Mar 16;16(3):e0248594. doi: 10.1371/journal.pone.0248594. eCollection 2021. PMID: 33725017
2020
Dinnyes A, Schnur A, Muenthaisong S, Bartenstein P, Burcez CT, Burton N, Cyran C, Gianello P, Kemter E, Nemeth G, Nicotra F, Prepost E, Qiu Y, Russo L, Wirth A, Wolf E, Ziegler S, Kobolak J. Integration of nano- and biotechnology for beta-cell and islet transplantation in type-1 diabetes treatment. Cell Prolif 2020; 53:e12785
Hofmann I, Kemter E, Theobalt N, Fiedler S, Bidlingmaier M, Hinrichs A, Aichler M, Burkhardt K, Klymiuk N, Wolf E, Wanke R, Blutke A. Linkage between growth retardation and pituitary cell morphology in a dystrophin-deficient pig model of Duchenne muscular dystrophy. Growth hormone & IGF research : official journal of the Growth Hormone Research Society and the International IGF Research Society 2020; 51:6-16
Ludwig B, Wolf E, Schoenmann U, Ludwig S. Large Animal Models of Diabetes. Methods in molecular biology 2020; 2128:115-134
Moretti A, Fonteyne L, Giesert F, Hoppmann P, Meier AB, Bozoglu T, Baehr A, Schneider CM, Sinnecker D, Klett K, Frohlich T, Rahman FA, Haufe T, Sun S, Jurisch V, Kessler B, Hinkel R, Dirschinger R, Martens E, Jilek C, Graf A, Krebs S, Santamaria G, Kurome M, Zakhartchenko V, Campbell B, Voelse K, Wolf A, Ziegler T, Reichert S, Lee S, Flenkenthaler F, Dorn T, Jeremias I, Blum H, Dendorfer A, Schnieke A, Krause S, Walter MC, Klymiuk N, Laugwitz KL, Wolf E, Wurst W, Kupatt C. Somatic gene editing ameliorates skeletal and cardiac muscle failure in pig and human models of Duchenne muscular dystrophy. Nat Med 2020; 26:207-214
Renner S, Blutke A, Clauss S, Deeg CA, Kemter E, Merkus D, Wanke R, Wolf E. Porcine models for studying complications and organ crosstalk in diabetes mellitus. Cell and tissue research 2020 doi: 10.1007/s00441-019-03158-9
Riedel EO, Hinrichs A, Kemter E, Dahlhoff M, Backman M, Rathkolb B, Prehn C, Adamski J, Renner S, Blutke A, Hrabĕ de Angelis M, Bidlingmaier M, Schopohl J, Arnold GJ, Fröhlich T, Wolf E. Functional changes of the liver in the absence of growth hormone (GH) action – proteomic and metabolomic insights from a GH receptor deficient pig model. Molecular metabolism 2020 doi: 10.1016/j.molmet.2020.100978
Tamiyakul H, Kemter E, Kösters M, Ebner S, Blutke A, Klymiuk N, Flenkenthaler F, Wolf E, Arnold GJ, Fröhlich T. Progressive Proteome Changes in the Myocardium of a Pig Model for Duchenne Muscular Dystrophy. iScience 2020; 23:101516
Tissières V, Geier F, Kessler B, Wolf E, Zeller R, Lopez-Rios J. Gene Regulatory and Expression Differences between Mouse and Pig Limb Buds Provide Insights into the Evolutionary Emergence of Artiodactyl Traits. Cell Rep. 2020 Apr 7;31(1):107490. doi: 10.1016/j.celrep.2020.03.054. PubMed PMID: 32268095.
Vohra T, Kemter E, Sun N, Dobenecker B, Hinrichs A, Burrello J, Gomez-Sanchez EP, Gomez-Sanchez CE, Wang J, Kinker IS, Teupser D, Fischer K, Schnieke A, Peitzsch M, Eisenhofer G, Walch A, Reincke M, Wolf E, Williams TA. Effect of Dietary Sodium Modulation on Pig Adrenal Steroidogenesis and Transcriptome Profiles. Hypertension 2020; 76:1769-1777
Weigand M, Degroote RL, Amann B, Renner S, Wolf E, Hauck SM, Deeg CA. Proteome profile of neutrophils from a transgenic diabetic pig model shows distinct changes. J Proteomics 2020; 224:103843
Zettler S, Renner S, Kemter E, Hinrichs A, Klymiuk N, Backman M, Riedel EO, Mueller C, Streckel E, Braun-Reichhart C, Martins AS, Kurome M, Keßler B, Zakhartchenko V, Flenkenthaler F, Arnold GJ, Fröhlich T, Blum H, Blutke A, Wanke R, Wolf E. A decade of experience with genetically tailored pig models for diabetes and metabolic research. Anim Reprod 2020; 17:e20200064
Zhao S, Todorov MI, Cai R, Maskari RA, Steinke H, Kemter E, Mai H, Rong Z, Warmer M, Stanic K, Schoppe O, Paetzold JC, Gesierich B, Wong MN, Huber TB, Duering M, Bruns OT, Menze B, Lipfert J, Puelles VG, Wolf E, Bechmann I, Ertürk A. Cellular and Molecular Probing of Intact Human Organs. Cell 2020; 180:796-812.e719
2019
Backman M, Flenkenthaler F, Blutke A, Dahlhoff M, Landstrom E, Renner S, Philippou-Massier J, Krebs S, Rathkolb B, Prehn C, Grzybek M, Coskun U, Rothe M, Adamski J, de Angelis MH, Wanke R, Fröhlich T, Arnold GJ, Blum H, Wolf E. Multi-omics insights into functional alterations of the liver in insulin-deficient diabetes mellitus. Molecular metabolism 2019; 26:30-44
Clauss S, Bleyer C, Schuttler D, Tomsits P, Renner S, Klymiuk N, Wakili R, Massberg S, Wolf E, Kaab S. Animal models of arrhythmia: classic electrophysiology to genetically modified large animals. Nature reviews Cardiology 2019; 16:457-475
Regensburger AP, Fonteyne LM, Jungert J, Wagner AL, Gerhalter T, Nagel AM, Heiss R, Flenkenthaler F, Qurashi M, Neurath MF, Klymiuk N, Kemter E, Frohlich T, Uder M, Woelfle J, Rascher W, Trollmann R, Wolf E, Waldner MJ, Knieling F. Detection of collagens by multispectral optoacoustic tomography as an imaging biomarker for Duchenne muscular dystrophy. Nat Med 2019; 25:1905-1915
Renner S, Martins AS, Streckel E, Braun-Reichhart C, Backman M, Prehn C, Klymiuk N, Baehr A, Blutke A, Landbrecht-Schessl C, Wuensch A, Kessler B, Kurome M, Hinrichs A, Koopmans SJ, Krebs S, Kemter E, Rathkolb B, Nagashima H, Blum H, Ritzmann M, Wanke R, Aigner B, Adamski J, Hrabe de Angelis M, Wolf E. Mild maternal hyperglycemia in INS (C93S) transgenic pigs causes impaired glucose tolerance and metabolic alterations in neonatal offspring. Disease models & mechanisms 2019; 12
2018
Hinrichs A, Kessler B, Kurome M, Blutke A, Kemter E, Bernau M, Scholz AM, Rathkolb B, Renner S, Bultmann S, Leonhardt H, de Angelis MH, Nagashima H, Hoeflich A, Blum WF, Bidlingmaier M, Wanke R, Dahlhoff M, Wolf E. Growth hormone receptor-deficient pigs resemble the pathophysiology of human Laron syndrome and reveal altered activation of signaling cascades in the liver. Molecular metabolism 2018; 11:113-128
Kleinert M, Clemmensen C, Hofmann SM, Moore MC, Renner S, Woods SC, Huypens P, Beckers J, de Angelis MH, Schurmann A, Bakhti M, Klingenspor M, Heiman M, Cherrington AD, Ristow M, Lickert H, Wolf E, Havel PJ, Muller TD, Tschop MH. Animal models of obesity and diabetes mellitus. Nature reviews Endocrinology 2018; 14:140-162
Matsunari H, Watanabe M, Nakano K, Enosawa S, Umeyama K, Uchikura A, Yashima S, Fukuda T, Klymiuk N, Kurome M, Kessler B, Wuensch A, Zakhartchenko V, Wolf E, Hanazono Y, Nagaya M, Umezawa A, Nakauchi H, Nagashima H. Modeling lethal X-linked genetic disorders in pigs with ensured fertility. Proceedings of the National Academy of Sciences of the United States of America 2018; 115:708-713
Renner S, Blutke A, Dobenecker B, Dhom G, Muller TD, Finan B, Clemmensen C, Bernau M, Novak I, Rathkolb B, Senf S, Zols S, Roth M, Gotz A, Hofmann SM, Hrabe de Angelis M, Wanke R, Kienzle E, Scholz AM, DiMarchi R, Ritzmann M, Tschop MH, Wolf E. Metabolic syndrome and extensive adipose tissue inflammation in morbidly obese Gottingen minipigs. Molecular metabolism 2018; 16:180-190
Vogel H, Kamitz A, Hallahan N, Lebek S, Schallschmidt T, Jonas W, Jahnert M, Gottmann P, Zellner L, Kanzleiter T, Damen M, Altenhofen D, Burkhardt R, Renner S, Dahlhoff M, Wolf E, Muller TD, Bluher M, Joost HG, Chadt A, Al-Hasani H, Schurmann A. A collective diabetes cross in combination with a computational framework to dissect the genetics of human obesity and Type 2 diabetes. Human molecular genetics 2018; 27:3099-3112
2017
Blutke A, Renner S, Flenkenthaler F, Backman M, Haesner S, Kemter E, Landstrom E, Braun-Reichhart C, Albl B, Streckel E, Rathkolb B, Prehn C, Palladini A, Grzybek M, Krebs S, Bauersachs S, Bahr A, Bruhschwein A, Deeg CA, De Monte E, Dmochewitz M, Eberle C, Emrich D, Fux R, Groth F, Gumbert S, Heitmann A, Hinrichs A, Kessler B, Kurome M, Leipig-Rudolph M, Matiasek K, Ozturk H, Otzdorff C, Reichenbach M, Reichenbach HD, Rieger A, Rieseberg B, Rosati M, Saucedo MN, Schleicher A, Schneider MR, Simmet K, Steinmetz J, Ubel N, Zehetmaier P, Jung A, Adamski J, Coskun U, Hrabe de Angelis M, Simmet C, Ritzmann M, Meyer-Lindenberg A, Blum H, Arnold GJ, Froehlich T, Wanke R, Wolf E. The Munich MIDY Pig Biobank - A unique resource for studying organ crosstalk in diabetes. Molecular metabolism 2017; 6:931-940
Eichhorn S, Mendoza A, Prinzing A, Stroh A, Xinghai L, Polski M, Heller M, Lahm H, Wolf E, Lange R, Krane M. Corpuls CPR Generates Higher Mean Arterial Pressure Than LUCAS II in a Pig Model of Cardiac Arrest. BioMed research international 2017; 2017:5470406
Hinkel R, Hoewe A, Renner S, Ng J, Lee S, Klett K, Kaczmarek V, Moretti A, Laugwitz KL, Skroblin P, Mayr M, Milting H, Dendorfer A, Reichart B, Wolf E, Kupatt C. Diabetes mellitus-induced microvascular destabilization in the myocardium. Journal of the American College of Cardiology 2017; 69:131-143
Kleinwort KJH, Amann B, Hauck SM, Hirmer S, Blutke A, Renner S, Uhl PB, Lutterberg K, Sekundo W, Wolf E, Deeg CA. Retinopathy with central oedema in an INS C94Y transgenic pig model of long-term diabetes. Diabetologia 2017; 60:1541-1549
2016
Albl B, Haesner S, Braun-Reichhart C, Streckel E, Renner S, Seeliger F, Wolf E, Wanke R, Blutke A. Tissue sampling guides for porcine biomedical models. Toxicologic pathology 2016; 44:414-420
Froehlich T, Kemter E, Flenkenthaler F, Klymiuk N, Otte KA, Blutke A, Krause S, Walter MC, Wanke R, Wolf E, Arnold GJ. Progressive muscle proteome changes in a clinically relevant pig model of Duchenne muscular dystrophy. Scientific reports 2016; 6:33362
Klymiuk N, Seeliger F, Bohlooly YM, Blutke A, Rudmann DG, Wolf E. Tailored pig models for preclinical efficacy and safety testing of targeted therapies. Toxicologic pathology 2016; 44:346-357
Renner S, Blutke A, Streckel E, Wanke R, Wolf E. Incretin actions and consequences of incretin-based therapies: lessons from complementary animal models. The Journal of pathology 2016a; 238:345-358
Renner S, Dobenecker B, Blutke A, Zols S, Wanke R, Ritzmann M, Wolf E. Comparative aspects of rodent and nonrodent animal models for mechanistic and translational diabetes research. Theriogenology 2016b; 86:406-421
Schneider MR, Wolf E. Genetically engineered pigs as investigative and translational models in dermatology. The British journal of dermatology 2016; 174:237-239
2015
Guillon A, Chevaleyre C, Barc C, Berri M, Adriaensen H, Lecompte F, Villemagne T, Pezant J, Delaunay R, Moenne-Loccoz J, Berthon P, Baehr A, Wolf E, Klymiuk N, Attucci S, Ramphal R, Sarradin P, Buzoni-Gatel D, Si-Tahar M, Caballero I. Computed tomography (CT) scanning facilitates early identification of neonatal cystic fibrosis piglets. PloS one 2015; 10:e0143459
Li S, Edlinger M, Saalfrank A, Flisikowski K, Tschukes A, Kurome M, Zakhartchenko V, Kessler B, Saur D, Kind A, Wolf E, Schnieke A, Flisikowska T. Viable pigs with a conditionally-activated oncogenic KRAS mutation. Transgenic research 2015; 24:509-517
Streckel E, Braun-Reichhart C, Herbach N, Dahlhoff M, Kessler B, Blutke A, Baehr A, Ubel N, Eddicks M, Ritzmann M, Krebs S, Goeke B, Blum H, Wanke R, Wolf E, Renner S. Effects of the glucagon-like peptide-1 receptor agonist liraglutide in juvenile transgenic pigs modeling a pre-diabetic condition. Journal of translational medicine 2015; 13:73
2014
Wolf E, Braun-Reichhart C, Streckel E, Renner S. Genetically engineered pig models for diabetes research. Transgenic research 2014; 23:27-38
2013
Klymiuk N, Blutke A, Graf A, Krause S, Burkhardt K, Wuensch A, Krebs S, Kessler B, Zakhartchenko V, Kurome M, Kemter E, Nagashima H, Schoser B, Herbach N, Blum H, Wanke R, Aartsma-Rus A, Thirion C, Lochmuller H, Walter MC, Wolf E. Dystrophin-deficient pigs provide new insights into the hierarchy of physiological derangements of dystrophic muscle. Human molecular genetics 2013; 22:4368-4382
Renner S, Braun-Reichhart C, Blutke A, Herbach N, Emrich D, Streckel E, Wunsch A, Kessler B, Kurome M, Bahr A, Klymiuk N, Krebs S, Puk O, Nagashima H, Graw J, Blum H, Wanke R, Wolf E. Permanent neonatal diabetes in INS(C94Y) transgenic pigs. Diabetes 2013; 62:1505-1511
2012
Baehr A, Wolf E. Domestic animal models for biomedical research. Reproduction in domestic animals = Zuchthygiene 2012; 47 Suppl 4:59-71
Flisikowska T, Merkl C, Landmann M, Eser S, Rezaei N, Cui X, Kurome M, Zakhartchenko V, Kessler B, Wieland H, Rottmann O, Schmid RM, Schneider G, Kind A, Wolf E, Saur D, Schnieke A. A porcine model of familial adenomatous polyposis. Gastroenterology 2012; 143:1173-1175 e1171-1177
Klymiuk N, Bocker W, Schonitzer V, Baehr A, Radic T, Froehlich T, Wuensch A, Kessler B, Kurome M, Schilling E, Herbach N, Wanke R, Nagashima H, Mutschler W, Arnold GJ, Schwinzer R, Schieker M, Wolf E. First inducible transgene expression in porcine large animal models. FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2012a; 26:1086-1099
Klymiuk N, Mundhenk L, Kraehe K, Wuensch A, Plog S, Emrich D, Langenmayer MC, Stehr M, Holzinger A, Kroner C, Richter A, Kessler B, Kurome M, Eddicks M, Nagashima H, Heinritzi K, Gruber AD, Wolf E. Sequential targeting of CFTR by BAC vectors generates a novel pig model of cystic fibrosis. Journal of molecular medicine 2012b; 90:597-608
Leuchs S, Saalfrank A, Merkl C, Flisikowska T, Edlinger M, Durkovic M, Rezaei N, Kurome M, Zakhartchenko V, Kessler B, Flisikowski K, Kind A, Wolf E, Schnieke A. Inactivation and inducible oncogenic mutation of p53 in gene targeted pigs. PloS one 2012; 7:e43323
Renner S, Romisch-Margl W, Prehn C, Krebs S, Adamski J, Goke B, Blum H, Suhre K, Roscher AA, Wolf E. Changing metabolic signatures of amino acids and lipids during the prediabetic period in a pig model with impaired incretin function and reduced beta-cell mass. Diabetes 2012; 61:2166-2175
Sax G, Kessler B, Wolf E, Winter G. In-vivo biodegradation of extruded lipid implants in rabbits. Journal of controlled release : official journal of the Controlled Release Society 2012; 163:195-202
Walters EM, Wolf E, Whyte JJ, Mao J, Renner S, Nagashima H, Kobayashi E, Zhao J, Wells KD, Critser JK, Riley LK, Prather RS. Completion of the swine genome will simplify the production of swine as a large animal biomedical model. BMC medical genomics 2012; 5:55
2010
Aigner B, Renner S, Kessler B, Klymiuk N, Kurome M, Wunsch A, Wolf E. Transgenic pigs as models for translational biomedical research. Journal of molecular medicine 2010b; 88:653-664
Renner S, Fehlings C, Herbach N, Hofmann A, von Waldthausen DC, Kessler B, Ulrichs K, Chodnevskaja I, Moskalenko V, Amselgruber W, Goke B, Pfeifer A, Wanke R, Wolf E. Glucose intolerance and reduced proliferation of pancreatic beta-cells in transgenic pigs with impaired glucose-dependent insulinotropic polypeptide function. Diabetes 2010; 59:1228-1238

